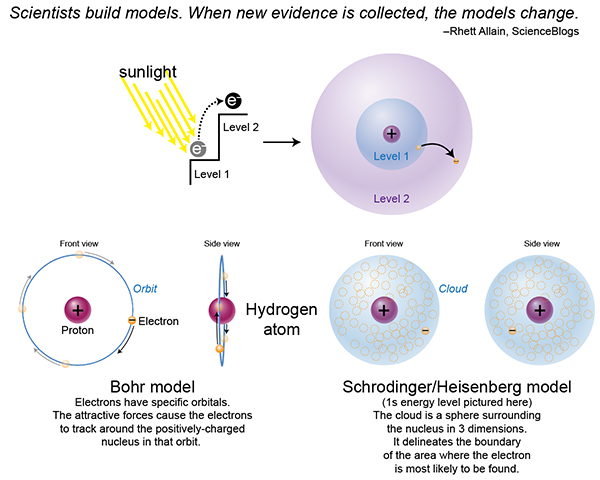
In the last post when I talked about exciting electrons to higher energy levels, it got me thinking about what a difficult topic that is, without first explaining where electrons are within an atom, and what influences its location. We were taught many years ago that electrons are the particles that carry a negative charge, and orbit the positively-charged particle (proton) because they are constantly being pulled toward that attractive force. This attractive force is also what keeps the electron from liberating itself entirely.
An infusion of energy–from sunlight for example–can allow the electron to temporarily overcome the attractive pull of that proton. The electron can now jump to an energy level of a higher-state, because it now possesses a higher level of energy.
Throughout the years, many atomic models have come and gone. The currently accepted model is the Schrodinger/Heisenberg, which dispels the idea that we can determine an electron’s discrete position. Rather, we can approximate the most likely region/s that an electron of a certain energy level can be found. This is called its probability distribution, and there are different probability distributions for different energy levels. So if an electron gets a boost of energy, and thereby enters a different energy level, it can go from one probability distribution to another, as dictated by that energy level.
Please folks, if anyone sees an error let me know!

 RSS Feed
RSS Feed
