As a last-gasp ode to summer, this topic–lead in beautifully from our mosquito talks–will be about grilling! We are going to unpack a few things about grilling, starting with how that smoky, charred, delicious flavor and smell are created. And we really can’t talk about that without our beloved chemistry.
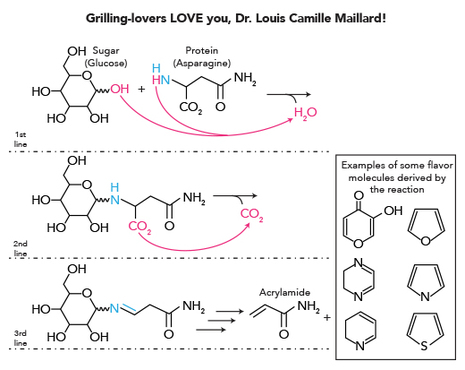
Since we discussed how mosquitoes use odor molecules to identify us as tasty morsels, we can expand on this with looking at a chemical reaction that is generally responsible for producing the odor molecules produced in grilling meat: the Maillard Reaction. There are additional products produced by this reaction: flavor, and the physical “browning” on the surface of the protein, which occurs when sugars react with proteins and amino acids as sufficient heat is applied.
To analyze this reaction a little more closely, in the first line you’ll see that there are 2 molecules, a sugar and a protein. In the 2nd line, now these two molecules are combined into one, and that atoms from each are lost, two hydrogen molecules and an oxygen (released as water). In the 2nd line, when a carbon dioxide is released, a double bond is formed in its place. Chemistry is very elegant because whatever is on the left of the arrow has to be equivalent to what is on the right.
Now comes to the grilling CONTROVERSY: we have heard about how charring/grilling/smoking can cause cancer. What in this process could be responsible for this? Hint, it's a possible product found in the 3rd line....
Since we discussed how mosquitoes use odor molecules to identify us as tasty morsels, we can expand on this with looking at a chemical reaction that is generally responsible for producing the odor molecules produced in grilling meat: the Maillard Reaction. There are additional products produced by this reaction: flavor, and the physical “browning” on the surface of the protein, which occurs when sugars react with proteins and amino acids as sufficient heat is applied.
To analyze this reaction a little more closely, in the first line you’ll see that there are 2 molecules, a sugar and a protein. In the 2nd line, now these two molecules are combined into one, and that atoms from each are lost, two hydrogen molecules and an oxygen (released as water). In the 2nd line, when a carbon dioxide is released, a double bond is formed in its place. Chemistry is very elegant because whatever is on the left of the arrow has to be equivalent to what is on the right.
Now comes to the grilling CONTROVERSY: we have heard about how charring/grilling/smoking can cause cancer. What in this process could be responsible for this? Hint, it's a possible product found in the 3rd line....

 RSS Feed
RSS Feed
