Now comes the FUN after these technical discussions! I guess fun is all relative....
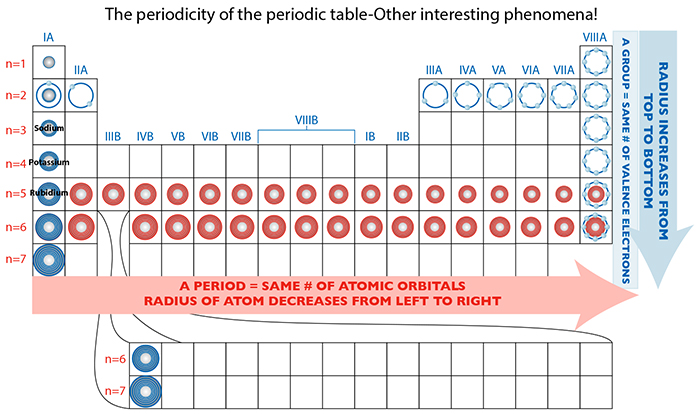
One of the patterns resulting from this organizational structure is that you intuitively think that the atom of an element must also physically grow in size if it’s adding electrons, right? Well, let’s take a closer look.
When we look at several elements in group IA, Sodium has 11 electrons, Potassium has 19, and Rubidium has 37. Each element has a significantly greater number of electrons than its preceding group member, and thus, requires an entirely new shell in which to house these electrons. Therefore, it increases by one orbital shell. And the atomic dimension does increase as a result.
When we look at a period, where we KNOW that each successive element has one more proton and one more electron (not discounting the neutron’s contribution to mass and size, but let’s simplify for this example), what we think might happen is the atom itself gets physically bigger at very tiny increments, since we’re adding in tiny increments.
In reality this is not the case! From left to right (e.g. n=5 and n=6), the atomic radius of each element decreases. Why? Answer, next post!
One of the patterns resulting from this organizational structure is that you intuitively think that the atom of an element must also physically grow in size if it’s adding electrons, right? Well, let’s take a closer look.
When we look at several elements in group IA, Sodium has 11 electrons, Potassium has 19, and Rubidium has 37. Each element has a significantly greater number of electrons than its preceding group member, and thus, requires an entirely new shell in which to house these electrons. Therefore, it increases by one orbital shell. And the atomic dimension does increase as a result.
When we look at a period, where we KNOW that each successive element has one more proton and one more electron (not discounting the neutron’s contribution to mass and size, but let’s simplify for this example), what we think might happen is the atom itself gets physically bigger at very tiny increments, since we’re adding in tiny increments.
In reality this is not the case! From left to right (e.g. n=5 and n=6), the atomic radius of each element decreases. Why? Answer, next post!

 RSS Feed
RSS Feed
