OK, back to our story on how our current atomic model came to be.
Rutherford's atomic model failed to describe electron movement. By laws of physics, electrons should lose energy when in continuous orbit, which then throws the entire atom into an unstable state. But we know the atom is very stable else it couldn’t be a building block.
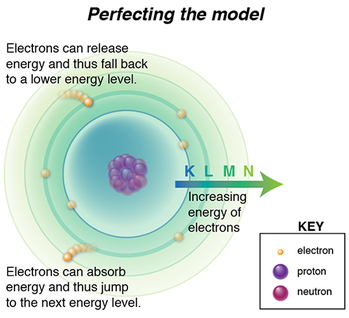
Bohr built upon Rutherford’s discovery of the nucleus, adding that an electron’s energy dictates the orbital in which it resides. So, electrons with higher energy are able to exist further away from the attractive force of the positively-charged nucleus, while that same attractive force exerted on lower-energy, weaker electrons keeps them closer to the nucleus.
The reason why this was such an important revelation is because it incorporated quantum mechanics to explain, even predict, electron movement. Furthermore, describing orbitals as distinct shells of energy meant knowing the gap from one energy level to the next. So if electrons gained and lost enough energy to hop between levels, those energy absorption and emission packets could be calculated. Data was being collected that described energy emissions, and wouldn’t you know? They matched Bohr’s calculations!
Sadly, orbitals are NOT exactly how electrons move about the atom nor can they accurately describe their location. So, although Bohr’s model took a huge leap forward, it’s still not quite right....
Rutherford's atomic model failed to describe electron movement. By laws of physics, electrons should lose energy when in continuous orbit, which then throws the entire atom into an unstable state. But we know the atom is very stable else it couldn’t be a building block.
Bohr built upon Rutherford’s discovery of the nucleus, adding that an electron’s energy dictates the orbital in which it resides. So, electrons with higher energy are able to exist further away from the attractive force of the positively-charged nucleus, while that same attractive force exerted on lower-energy, weaker electrons keeps them closer to the nucleus.
The reason why this was such an important revelation is because it incorporated quantum mechanics to explain, even predict, electron movement. Furthermore, describing orbitals as distinct shells of energy meant knowing the gap from one energy level to the next. So if electrons gained and lost enough energy to hop between levels, those energy absorption and emission packets could be calculated. Data was being collected that described energy emissions, and wouldn’t you know? They matched Bohr’s calculations!
Sadly, orbitals are NOT exactly how electrons move about the atom nor can they accurately describe their location. So, although Bohr’s model took a huge leap forward, it’s still not quite right....

 RSS Feed
RSS Feed
