We’ve now "completed" the history of the development of the atomic model with Heisenberg. With his clarification that electron orbitals aren’t so much a trajectory or travel path, but rather a cloud of probability in which a single electron can be found, let’s apply THIS model to NaCl, instead of using the incorrect-but-easier-to-understand Bohr model.
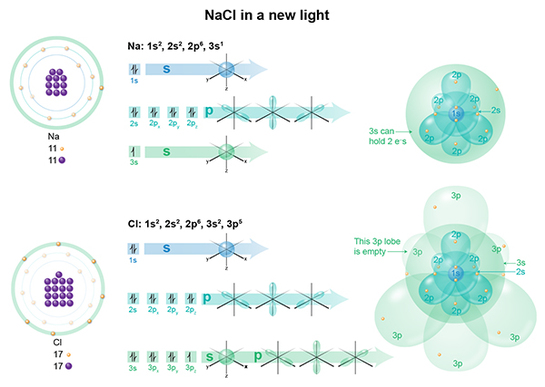
This image shows the Bohr model of each atom on the left, its electron configuration in the middle, and then its application of the electron cloud model on right. I think you can immediately see why Bohr’s model, for certain atoms, is much easier to teach with, than the cloud model on right. But let’s walk through this.
In a Na atom, we have 11 electrons, so if we fill the clouds one by one following Aufbau's principle, we get 1s2 and 2s2 (spherical clouds), 2p6 (dumb bell cloud), and 3s1 (spherical cloud). This last cloud is only half-filled, as 3s2 can hold 2 electrons.
In a Cl atom, we have 17 electrons, so if we fill the clouds one by one, we get 1s2 and 2s2 (spherical clouds), 2p6 (dumbell cloud), 3s2 (spherical cloud) and 3p5 (dumbell cloud). This last cloud is almost filled, as 3p6 can hold 6 electrons. There's one lone lobe BEGGING to be filled, while in Na, there's one lonely electron.
What would happen if that electron were to pick up and move to the Cl suburb?
This image shows the Bohr model of each atom on the left, its electron configuration in the middle, and then its application of the electron cloud model on right. I think you can immediately see why Bohr’s model, for certain atoms, is much easier to teach with, than the cloud model on right. But let’s walk through this.
In a Na atom, we have 11 electrons, so if we fill the clouds one by one following Aufbau's principle, we get 1s2 and 2s2 (spherical clouds), 2p6 (dumb bell cloud), and 3s1 (spherical cloud). This last cloud is only half-filled, as 3s2 can hold 2 electrons.
In a Cl atom, we have 17 electrons, so if we fill the clouds one by one, we get 1s2 and 2s2 (spherical clouds), 2p6 (dumbell cloud), 3s2 (spherical cloud) and 3p5 (dumbell cloud). This last cloud is almost filled, as 3p6 can hold 6 electrons. There's one lone lobe BEGGING to be filled, while in Na, there's one lonely electron.
What would happen if that electron were to pick up and move to the Cl suburb?

 RSS Feed
RSS Feed
