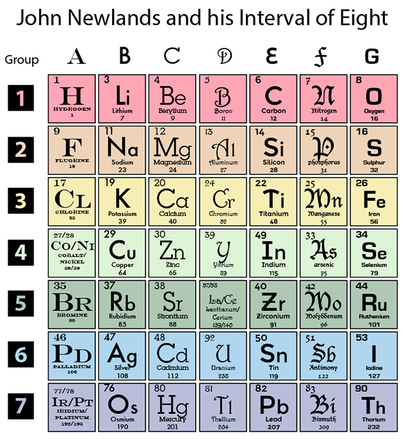
After de Chancourtois’ little known elemental organizational proposal, a chemist named John Newlands was expanding upon Dobreiner’s Law of Triad to a Law of Octaves, or 8-repeating sequences. He thus grouped them in a manner similar to the grid you see here (I couldn't find one figure to confirm what the original 56-element grid might have been). However, variation in the versions among the 5-6 references I came across included elements like Di, Ro, G, Gl and Ds....none of which exist today. Perhaps they were placeholders for undiscovered/unnamed elements? Possibly.
The key concept to take away here is that Newlands began to see similarities in physical properties having atomic weights close in range that came with groups of 8. So, the rows show the groupings based on relatively close atomic weights (designated by color). But, he also noted similarities in reactive, or chemical properties. So for these elements, he stacked these elements to form columns (designated by font), to denote that the element forms a second grouping, aside from the rows which assigned it to a first grouping.
Each element is now categorized in 2 ways: a row, or Periodic, grouping, and a column, or Group, grouping. Herein lies the foundation of the Periodic Table!
The key concept to take away here is that Newlands began to see similarities in physical properties having atomic weights close in range that came with groups of 8. So, the rows show the groupings based on relatively close atomic weights (designated by color). But, he also noted similarities in reactive, or chemical properties. So for these elements, he stacked these elements to form columns (designated by font), to denote that the element forms a second grouping, aside from the rows which assigned it to a first grouping.
Each element is now categorized in 2 ways: a row, or Periodic, grouping, and a column, or Group, grouping. Herein lies the foundation of the Periodic Table!

 RSS Feed
RSS Feed
