Since I so conveniently steered our conversation back to chemistry by talking about atomic models, I thought I’d take a stroll down memory lane on how our current model of the atom developed. This also demonstrates how scientists constantly re-evaluate theory in the face of new evidence. If this is NOT the ultimate example of keeping an open mind, I don’t know what is.
We can go way back the 500’s BC to find theories developing on the concept of atoms. To streamline, let’s start with John Dalton, the first scientist to truly organize elements using the characteristics those of atoms. By experimenting with numerous gases, he observed that the elements that made up the chemical combined in specific ratios. He deduced this by weighing their individual masses. Further employing deductive reasoning, he put together “rules” to describe/predict how elements combine to form chemicals and more specifically, that they combine in specific ratios.
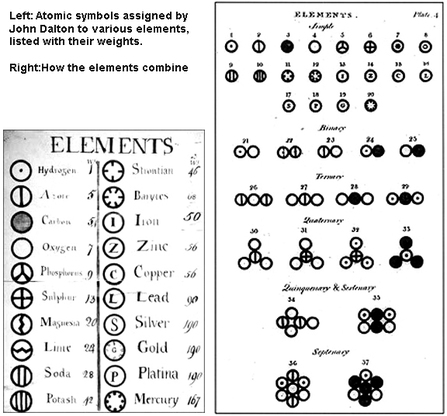
Rather than drawing my own illustration of Dalton’s Atomic model, I felt these original images of Dalton’s would be more impactful to begin our journey. More than a simple list of various elements, Mr. Dalton begins to notice patterns of "behavior" with certain groups of elements, and thus organizes them visually by these characteristics. This will become the prototype for the periodic table.
We can go way back the 500’s BC to find theories developing on the concept of atoms. To streamline, let’s start with John Dalton, the first scientist to truly organize elements using the characteristics those of atoms. By experimenting with numerous gases, he observed that the elements that made up the chemical combined in specific ratios. He deduced this by weighing their individual masses. Further employing deductive reasoning, he put together “rules” to describe/predict how elements combine to form chemicals and more specifically, that they combine in specific ratios.
Rather than drawing my own illustration of Dalton’s Atomic model, I felt these original images of Dalton’s would be more impactful to begin our journey. More than a simple list of various elements, Mr. Dalton begins to notice patterns of "behavior" with certain groups of elements, and thus organizes them visually by these characteristics. This will become the prototype for the periodic table.

 RSS Feed
RSS Feed
