At last, I’m ready to unleash the history of the development of the periodic chart!
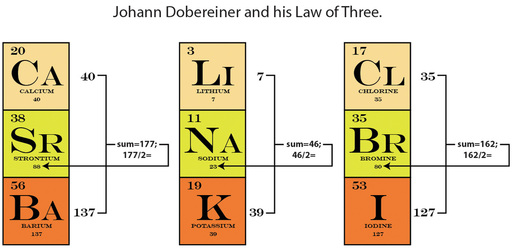
Picture it: the year is 1829, and a scientist named Johann Dobereiner begins to notice some interesting patterns occurring within elements grouped in threes. For instance, the atomic weight of the element Strontium falls neatly in between Calcium and Barium:
Ca (AW=40) + Ba (AW 137) = 177 divided by 2 = 88 = Strontium's Atomic Weight!
He recognized these patterns in other triad sets, and this is how the race to organize the elements as they were discovered began. By identifying patterns found in their physical and chemical properties, scientists were able to predict that those elements would then have similar behavioral properties. And to make sense of these similarities, scientists then began arranging elements accordingly.
Next, you likely won’t guess in which direction elemental organization went. HINT: Do NOT (yet) look to the current periodic table for a hint!
Picture it: the year is 1829, and a scientist named Johann Dobereiner begins to notice some interesting patterns occurring within elements grouped in threes. For instance, the atomic weight of the element Strontium falls neatly in between Calcium and Barium:
Ca (AW=40) + Ba (AW 137) = 177 divided by 2 = 88 = Strontium's Atomic Weight!
He recognized these patterns in other triad sets, and this is how the race to organize the elements as they were discovered began. By identifying patterns found in their physical and chemical properties, scientists were able to predict that those elements would then have similar behavioral properties. And to make sense of these similarities, scientists then began arranging elements accordingly.
Next, you likely won’t guess in which direction elemental organization went. HINT: Do NOT (yet) look to the current periodic table for a hint!

 RSS Feed
RSS Feed
