We begin to see the brilliance of all the groundbreaking work of all the scientists who came before. It may look like a simple grid filled with letters, but it’s so much more than that.
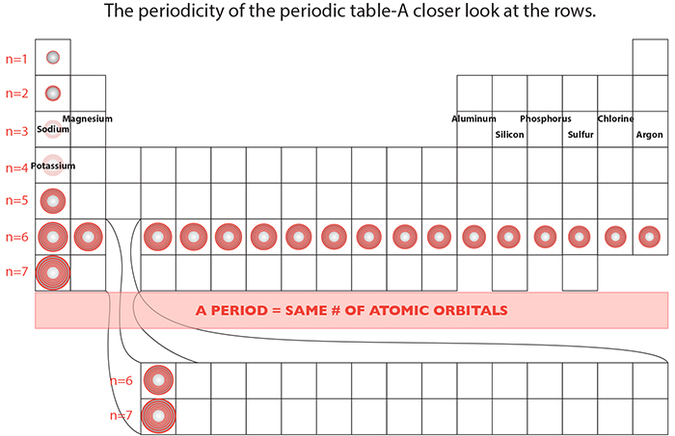
The main patterns run across the table in left-to-right rows and up-to-down in columns. The rows are called periods, and stress the periodicity of the table, in that periodically, an element suddenly exhibits similar chemical properties to that of a previous element. For instance, Sodium is in the same period as Magnesium, Aluminum, Silicon, through to Argon. Then we get to Potassium, and boom! Two things happen: 1) Now Potassium has four atomic orbitals instead of three, the common factor in the previous period of elements. But even with this major physical change that forces us to start a new period and naturally forming a column with Sodium, chemically it happens to BEHAVE like Sodium! Therefore, each column, or group, has a pattern as well.
Each period is thus defined by the number of atomic orbitals that those elements possess. The red number to the left of each period indicates its “n” or orbital shell level number. So, if n=3, then each element in that period utilizes up to the 3rd level of atomic orbital shells. The shell itself may not be full, but the number of shells with occupying electrons is the same.
The main patterns run across the table in left-to-right rows and up-to-down in columns. The rows are called periods, and stress the periodicity of the table, in that periodically, an element suddenly exhibits similar chemical properties to that of a previous element. For instance, Sodium is in the same period as Magnesium, Aluminum, Silicon, through to Argon. Then we get to Potassium, and boom! Two things happen: 1) Now Potassium has four atomic orbitals instead of three, the common factor in the previous period of elements. But even with this major physical change that forces us to start a new period and naturally forming a column with Sodium, chemically it happens to BEHAVE like Sodium! Therefore, each column, or group, has a pattern as well.
Each period is thus defined by the number of atomic orbitals that those elements possess. The red number to the left of each period indicates its “n” or orbital shell level number. So, if n=3, then each element in that period utilizes up to the 3rd level of atomic orbital shells. The shell itself may not be full, but the number of shells with occupying electrons is the same.

 RSS Feed
RSS Feed
