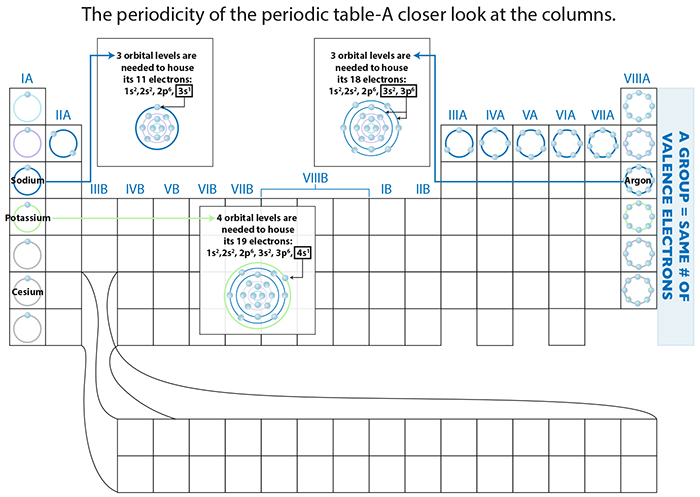
Next, let’s get back to our story of the periodic table, and look at the columns, called groups. An element within a group has the same number of VALENCE electrons, or as we learned before, the same number of electrons in whatever its outermost shell is. For example, look at group 8A where all the elements have 8 valence electrons. The difference between a group and the period is that we are talking about number of outermost electrons for each element in a group, versus the number of orbitals that house ALL of the electrons for each element in a period. In other words, with Sodium, we have 11 electrons, so, we need to use a spot in the 3s orbital to house the 11th electron. If you look at Argon, it too needs to utilize that 3rd orbital to house its last 8 of 18 electrons. Sodium needs only one slot for that 11th electron, versus Argon which needs to use 8 slots in the 3rd level orbital. However, both need to utilize that 3rd orbital shell.
Potassium has one more electron than Argon, and that is enough to bump that electron to the 4th level orbital shell. Potassium now differs from the previous period because it uses a 4th orbital shell. Yet it also has something in common with its group members...its valence electron number!
Remember, valence electron number is NOT the same as the total number of electrons for that element. For example, Sodium has a total of 11 electrons, but only 1 valence electron. Likewise, Cesium has only 1 valence electron, but in total has 55 electrons! Their commonality is having only 1 valence electron, and hence they react the same way chemically. And THAT is why they are group buddies.
Potassium has one more electron than Argon, and that is enough to bump that electron to the 4th level orbital shell. Potassium now differs from the previous period because it uses a 4th orbital shell. Yet it also has something in common with its group members...its valence electron number!
Remember, valence electron number is NOT the same as the total number of electrons for that element. For example, Sodium has a total of 11 electrons, but only 1 valence electron. Likewise, Cesium has only 1 valence electron, but in total has 55 electrons! Their commonality is having only 1 valence electron, and hence they react the same way chemically. And THAT is why they are group buddies.

 RSS Feed
RSS Feed
