Before we get to the Periodic Table discussions, let's make a few more points about electrons.
Louis de Broglie was the first scientist to deduce that an electron has both particle and wave properties. The electron's minute size IS its defining characteristic, and why we cannot define both its momentum and exact position in space simultaneously.
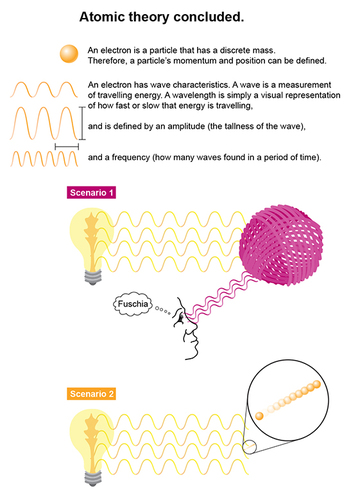
If we humans try to SEE where a ball of yarn (see scenario 1), for instance, is, we can’t see it unless we turn on a light. When the light waves reach the ball of yarn, they bounce off that ball and reflect back to our eye, which then perceives the color and shape of the ball. The yarn ball is unaffected.
With a teeny subatomic particle like the electron, turning on that light sends this wave of energy that affects that electron’s trajectory (scenario 2), and hence its momentum and position. Though this “observer effect” theory is separate from the Heisenberg Uncertainty Principle–which states that both position and momentum potential cannot be calculated simultaneously–it helps to explain why this is so. The electron can receive an energy transfer from the light wave of the light we use to see it, which then affects its momentum potential. Likewise, the light wave's force can change the electron’s position.
What does this ultimately mean? It means that any discrete “orbit” that previous atomic models have used to define location cannot be true! And thus, the idea of electron probability clouds replaced the electron orbit theory. Final words about those clouds next post!
Louis de Broglie was the first scientist to deduce that an electron has both particle and wave properties. The electron's minute size IS its defining characteristic, and why we cannot define both its momentum and exact position in space simultaneously.
If we humans try to SEE where a ball of yarn (see scenario 1), for instance, is, we can’t see it unless we turn on a light. When the light waves reach the ball of yarn, they bounce off that ball and reflect back to our eye, which then perceives the color and shape of the ball. The yarn ball is unaffected.
With a teeny subatomic particle like the electron, turning on that light sends this wave of energy that affects that electron’s trajectory (scenario 2), and hence its momentum and position. Though this “observer effect” theory is separate from the Heisenberg Uncertainty Principle–which states that both position and momentum potential cannot be calculated simultaneously–it helps to explain why this is so. The electron can receive an energy transfer from the light wave of the light we use to see it, which then affects its momentum potential. Likewise, the light wave's force can change the electron’s position.
What does this ultimately mean? It means that any discrete “orbit” that previous atomic models have used to define location cannot be true! And thus, the idea of electron probability clouds replaced the electron orbit theory. Final words about those clouds next post!

 RSS Feed
RSS Feed
