WHY are we spending SO much time on electrons? You may be secretly begging me to cease, but I cannot, because electrons are the essential reactive particle of the atom. Whenever a compound is formed, like salt (NaCl), the two elements, Na and Cl, are bound together because of electrons! Thank you chemistry for making my food taste delicious.
Where the electrons are within the spatial dimension of the atom, and how much energy they do or do not possess, are at the heart of all chemical reactions. This is why we are making such an effort to figure out where the electrons are and how much energy they possess. These facts are key to understanding why some elements react more readily with others, why different kinds of bonds form between elements, and, why some chemicals undergo changes with additions of energy (in the form of heat), or with other chemicals. It’s all about the electrons.
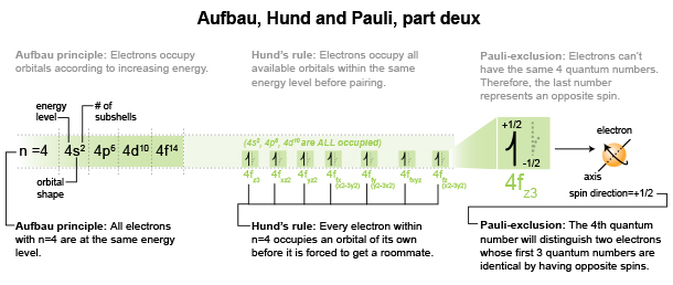
So, to look at Aufbau, Hund and Pauli more closely, I’ve isolated one energy level in the previous graphic for us to scrutinize. Next, we’ll go onto looking at some mind boggling sub-shells.
Where the electrons are within the spatial dimension of the atom, and how much energy they do or do not possess, are at the heart of all chemical reactions. This is why we are making such an effort to figure out where the electrons are and how much energy they possess. These facts are key to understanding why some elements react more readily with others, why different kinds of bonds form between elements, and, why some chemicals undergo changes with additions of energy (in the form of heat), or with other chemicals. It’s all about the electrons.
So, to look at Aufbau, Hund and Pauli more closely, I’ve isolated one energy level in the previous graphic for us to scrutinize. Next, we’ll go onto looking at some mind boggling sub-shells.

 RSS Feed
RSS Feed
